تأثير الزّراعة العضويّة على التّركيبِ الكيميائي والفعالية المضادّة للأكسدة للزيت العطري لنبات اكليل الجبل Rosmarinus officinalis L.
2025-08-23 | المجلد السابع العدد السادس - المجلد السابع | مقالات بحثيةعبير عبدالله مرضعه | جلال فندي | ميس رزق خازم
الملخص
خلفية البحث وهدفه: يعدّ نبات اكليل الجبل من النباتاتِ الطّبيّة والعطريّة ذات الأهميّة الصيدلانيّة الكبيرة، ويعزى ذلك بشكل رئيسي إلى زيته العطري الغني بالعديد من المركبات الفعالة بيولوجيّاً ذات الخصائص العلاجيّة الهامة كالتأثير المضاد للأكسدة. يتأثر التّركيب الكيميائي لهذا الزيت العطري بالعديد من العوامل كالأصل الجغرافي، المناخ، وأساليب الزراعة المتبعة. هدفت هذه الدّراسة إلى مقارنة تأثير الزراعة العضويّة والتّقليديّة على مردود الزيت العطري، تركيبه الكيميائي وفعاليته المضادّة للأكسدة.
مواد البحث وطرائقه: تمّ استحصال الزيت العطري من كُلّ عينة نباتيّة بطريقة التقطير المائي، وحُدّد تركيبها الكيميائي باستخدام جهاز الاستشراب الغازي المقترن بمتحري مطياف الكتلة GC-MS، كما تمّ تقييم الفعاليّة المضادّة للأكسدة باستخدام الجذر الحر DPPH• ضمن الطبق الميكروي ذو 96 بئر.
النتائج: أظهرت نتائج GC-MS أنّ الزيت العطري المستحصل من نبات اكليل الجبل المزروع زراعة عضويّة احتوى على نسب أعلى من 1,8-سينيول (38.96 %) والكافور (17.08 %)، في حين تميّز الزيت العطري المستحصل من نبات اكليل الجبل المزروع زراعة تقليديّة بنسبة أعلى من α-بينين (27.6 %) ونسبة أقل من 1,8-سينيول (15.26 %). أظهرت العينة العضويّة فعاليّة مضادّة للأكسدة (IC50 = 0.429 ± 0.006 ml/ml) متقاربة نوعاً ما مع العينة التّقليديّة (IC50 = 0.447 ± 0.002 ml/ml).
الاستنتاجات: أظهرت الزّراعة العضويّة تحسناً في مردود الزّيت العطري وارتفاعاً في نسب بعض المركبات الكيميائيّة الفعالة بيولوجيّاً في نبات اكليل الجبل مقارنة بالزراعة التّقليديّة، ممّا يشير إلى تحسّن في جودة الزيت العطري. بالمقابل، كانت الفعاليّة المضادّة للأكسدة متقاربة بين طريقتي الزراعة ولم تسجل فروقاً معنويّة.
كلمات مفتاحية : اكليل الجبل، عضوي، تقليدي، زيت عطري، GC-MS، مضاد أكسدة ,Rosmarinus officinalis, Organic, Conventional, Essential oil, GC-MS, Antioxidant
Introduction
Rosemary (Rosmarinus officinalis L.) is an aromatic, evergreen shrub from the Lamiaceae family, reaching approximately 1m in height. It features upright stems, dark green leaves, and whitish-blue flowers [1]. It is native Mediterranean range and cultivated in all parts of the world as a medicinal, culinary, and ornamental plant [2].
Rosmarinus officinalis essential oil (REO) secreted by glandular trichomes is mainly located in leaves and the flowers; the highest quality essential oil is obtained from the leaves [3]. REO contains many important bioactive compounds, such as monoterpene hydrocarbons (camphene, verbenene, α-pinene, β-pinene, limonene), oxygenated monoterpenes (1,8-cineol, linalool, camphor, borneol, verbenone) and sesquiterpenes (β-caryophyllene) [4] and it has been widely used for centuries as an ingredient in cosmetics, deodorants, soaps, perfumes, both for flavoring and for reservation of food products [5]. REO is utilized in aromatherapy as a nerve stimulant, particularly for improving memory and reducing lethargy. Studies have shown that when inhaled or applied through massage, REO significantly increases blood pressure, heart rate, and respiratory rate in humans. Furthermore, oral administration of REO has been found to elevate blood pressure in hypotensive patients. Inhalation of rosemary oil has also demonstrated benefits in enhancing concentration abilities [6]. Besides its psychostimulatory effects, REO possesses anticholinesterase [7], acaricidal [8], antibacterial, antioxidant [9], and antinociceptive properties [10].
The phytochemicals differ in quality and quantity depending on several factors such as the season of harvest, ecotypes, drying techniques, cultivar types, chemotypes, climatic conditions, soil, agricultural factors, extraction methods, and others [11][12].
Organic agriculture, also referred to as biological or ecological farming, integrates time-tested sustainable practices with advancements in modern technology. This approach prioritizes crop rotation, biological pest management, and the preservation of biodiversity within both plant and animal systems. Soil fertility is maintained through the application of compost, animal manure, and green manure [13], while the use of synthetic chemicals such as pesticides, antibiotics, genetic engineering, hormones, synthetic fertilizers, and irradiation is deliberately avoided [14][15].
Since the 1970s, this system has gained momentum due to growing concerns about the health and environmental risks associated with industrial farming [16].
In contrast, conventional agriculture is increasingly criticized for contributing to environmental degradation, climate change, and biodiversity loss [17].
The excessive generation of free radicals and lipid peroxidation in cellular membranes have been implicated in the pathogenesis of numerous disorders, such as cardiovascular diseases, genetic mutations, diabetes mellitus, ischemia-reperfusion injury, coronary atherosclerosis, Alzheimer’s disease, cancer development, and the aging process [18]. Recently, growing attention has been directed toward natural antioxidants derived from medicinal plants, owing to their favorable safety profile and potential health-promoting properties [19].
This study aimed to compare the chemical composition and antioxidant activity of essential oil of two samples of R. officinalis (organic cultivation and conventional cultivation).
Materials and Methods
- Plant material.
The aerial parts of R. officinalis (samples grown in conventional and organic cultivation) were collected in May 2023 from the same area (Dreikeesh – Rif Tartous, Syria, at attitude of 550 m, with coordinates 34˚53’34˚N 36˚7’6˚E) in order to exclude the environmental influence on the chemical composition, during the optimal harvesting period (the flowering season). The aerial parts of all samples were dried in the shade at room temperature.
- Essential Oil Distillation.
The essential oil was obtained from 100 g of plant aerial parts by Hydrodistillation for 4h using a Dean- Stark apparatus. The extracted oil was dried with anhydrous sodium sulfate. Both essential oil samples were stored at -5 ˚C in dry dark glass vials before use.
- Chromatographic Analyses of Essential Oils.
For the detection and quantitative determination of compounds of EOs, gas chromatography – mass spectrometry methods (GC-MS) were used. The carrier gas was helium with a flow rate of 1 mL/min, and the capillary columns used were HP-5MS 5% Phenyl Methyl Silox (30 m x 250 µm x 0.25 µm), split ratio of 8:1, the temperature program was (40 ˚C for 2 min, then 2.5 ˚C/min to 70 ˚C for 0 min, then 2 ˚C/min to 130 ˚C for 2 min, then 2.5 ˚C/min to 160 ˚C for 0 min, then 10 ˚C/min to 260 ˚C for 1 min), Injected quantity: 0.5 µl. The EO components were identified by comparing their spectra with the mass spectral database in the NIST library.
- Evaluation of Free Radical Scavenging Activity (RSA).
The DPPH• Radical Scavenging Activity was evaluated according to the Cheung et al method with some modifications by Choi et al [20].
For Rosemary EOs. Mixed 160 µl of DPPH• in methanol (0.2 mM) with 40 µl of the samples, or blank (methanol) in a 96-well microplate. The mixtures were left to stand at room temperature for 30 min, then the absorbance was measured at 517 nm by a microplate reader.
Free radical scavenging activity (RSA) was determined by the equation:
RSA (%) = (A0 – AS / A0) × 100
Where:
A0: Absorption of DPPH• solution.
AS: Absorption of DPPH• solution after 30 min of the sample addition.
Ascorbic acid determination curve. The calibration curve of ascorbic acid (from Panreac) was established with six dilutions of ascorbic acid standard (53 µg/ml) at concentrations of (5.3, 10.6, 21.2, 31.8, 42.4, 53) µg/mL. Then the previous reaction conditions were applied and the absorbance was read at 517 nm using the microplate reader, then the RSA was determined.
Gallic acid determination curve.
The calibration curve of gallic acid was established with six dilutions of gallic acid standard (120 µg/ml) at concentrations of (3, 6, 9, 12, 15, 18) µg/ml. Then the previous reaction conditions were applied and the absorbance was read at 517 nm using the microplate reader, then the RSA was determined.
Statistical Analysis.
All measurements were taken in triplicate, and values were then presented as average values along with their standard deviations (mean ± SD).
Results and Discussion
Yield of Essential Oils.
The oil extracted by hydro-distillation from the aerial parts of two different rosemary samples was a pale-yellow liquid, and had a characteristic odor. A high percentage yield of EO was obtained for the organically grown rosemary (1.6 ml/100g dry aerial parts), compared to the conventionally grown rosemary (1.4 ml/100g dry aerial parts).
Chemical Composition of Essential Oils.
The composition percentages of the EOs of organically and conventional rosemary are shown in (Table 1). The EO of organically grown samples contained 16 chemical constituents, making up 99.23 % of the total oil, whereas conventionally grown samples had 28 constituents, comprising 98.4 % of the oil. In the oil obtained from organically grown R. officinalis, oxygenated monoterpenes represented the main component at 71.87 %, followed by monoterpene hydrocarbons comprising 23.89 %. Also, analysis of the oil derived from conventionally grown R. officinalis showed that oxygenated monoterpenes were the major constituents at 57.26 %, with monoterpene hydrocarbons following at 39.14 %. Oxygenated compounds contribute more significantly than hydrocarbons to the fragrance and therapeutic properties.
| Table (1): Chemical composition of the Essential oils of Rosmarinus officinalis | |||||
|
pick no. |
compounds |
RT1
|
RT2 |
Percentage % | |
| -1-
Rosmarinus officinalis (Organic) |
-2-
Rosmarinus officinalis (Conventional) |
||||
| 1 | Tricyclene | – | 8.9446 | – | 0.1831 |
| 2 | α-pinene | 9.5306 | 9.5651 | 9.0123 | 27.9596 |
| 3 | Camphene | 10.1649 | 10.1788 | 13.9125 | 6.3914 |
| 4 | Bicyclo [3.1.0] hex-2-ene-4-methylene-1-(-1-methyl ethyl) | – | 10.5029 | – | 0.6226 |
| 5 | β-thujene | – | 10.8959 | – | 0.2839 |
| 6 | β-pinene | 11.5302 | 11.5372 | 0.3998 | 0.211 |
| 7 | α-myrcene | 12.0818 | – | 0.1939 | – |
| 8 | β-myrcene | – | 12.5577 | – | 1.846 |
| 9 | Delta-3-carene | – | 13.4334 | – | 0.3844 |
| 10 | α-terpinene | 13.7919 | 13.7506 | 0.1794 | 0.6073 |
| 11 | Cymene | – | 14.2195 | – | 0.4103 |
| 12 | p-cymene | – | 14.2609 | – | 0.8657 |
| 13 | 1,8-cineol | 14.5435 | 14.4608 | 38.9587 | 15.2563 |
| 14 | λ-terpinene | 16.157 | – | 0.1876 | – |
| 15 | Thujone | – | 18.8048 | – | 0.1639 |
| 16 | β-linalool | 19.1287 | 19.0117 | 0.2227 | 1.7307 |
| 17 | Camphor | 21.1699 | 21.1079 | 17.0829 | 9.4436 |
| 18 | Thujen-3-ol (Sabinol) | – | 21.5837 | – | 0.1579 |
| 19 | Pinocamphone | – | 22.1284 | – | 1.1852 |
| 20 | Borneol (Linderol) | 22.7696 | 22.7421 | 8.159 | 7.4381 |
| 21 | Iso-pinocamphone | – | 22.9972 | – | 0.8971 |
| 22 | 4-terpineol | 23.4591 | 23.4523 | 0.7294 | 0.465 |
| 23 | α-terpineol | 24.5003 | 24.5762 | 1.3382 | 1.4052 |
| 24 | Verbenone | – | 25.4933 | – | 12.2536 |
| 25 | Butyric acid-hexyl ester | – | 27.2585 | – | 0.7062 |
| 26 | Safranal | – | 27.6102 | – | 0.8886 |
| 27 | Bornyl acetate | 30.0441 | 30.0304 | 5.3742 | 5.7636 |
| 28 | Terpinene acetate | – | 35.3536 | – | 0.2104 |
| 29 | β-caryophyllene | 37.8772 | 37.8704 | 2.8014 | 0.3833 |
| 30 | α-caryophyllene | 39.9458 | – | 0.2978 | – |
| 31 | Delta-cadinene | 44.2829 | – | 0.3763 | – |
| 32 | Cis-α-copaene-8-ol | – | 51.3438 | – | 0.283 |
| Total of Identified Compounds | 99.23 | 98.40 | |||
| Monoterpene Hydrocarbons | 23.89 | 39.14 | |||
| Oxygenated Monoterpene | 71.87 | 57.26 | |||
| Sesquiterpene Hydrocarbons | 3.48 | 0.38 | |||
| Oxygenated Sesquiterpene | – | 0.28 | |||
|
RT1: retention time of components of EO of organic R. officinalis, RT2: retention time of components of EO of conventional R. officinalis |
|||||
In the essential oil of organically grown rosemary, three compounds were dominant: 1,8 – cineole (38.95%), camphor (17.08%), and camphene (13.91%). They were followed by α-pinene (9.01%), borneol (8.16%), bornyl acetate (5.37%), β-caryophyllene (2.8%), and α-terpineol (1.34%) (Figure 1) (Table 2).
|
Table (2): Comparative percentages of major bioactive compounds in essential oils from organically and conventionally cultivated R. officinalis |
||
| Percentage % | Compound | |
| Rosmarinus officinalis
(Conventional) |
Rosmarinus officinalis
(Organic) |
|
| 27.9596 | 9.0123 | α-pinene |
| 6.3914 | 13.9125 | Camphene |
| 15.2563 | 38.9587 | 1,8-cineol |
| 17.0829 | 9.4436 | Camphor |
| 8.159 | 7.4381 | Borneol |
| 5.3742 | 5.7636 | Bornyl acetate |
| 12 | 0 | Verbenone |
|
0 |
2.8014 | β-caryophyllene |
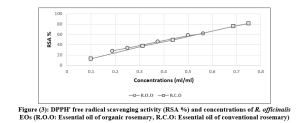
While, in the essential oil of conventional rosemary, three compounds were dominant: α-pinene (27.96%), 1,8 – cineole (15.26%), and verbenone (12.25%). They were followed by camphor (9.44%), borneol (7.44%), camphene (6.39%), bornyl acetate (5.76%), β-myrcene (1.8%), β-linalool (1.73%), α-terpineol (1.4%), and pinocamphone (1.2%) (Figure 2) (Table 2).
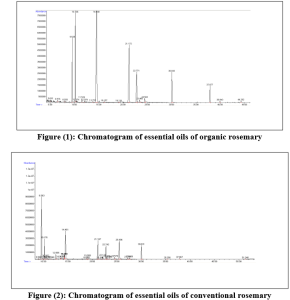
1,8 – cineole (Eucalyptol) exhibits a variety of biological activities, including anti-inflammatory, antioxidant, antimicrobial, bronchodilatory, antivirus, analgesic and pro-apoptotic effects. Recent evidence has also indicated its potential role in managing conditions such as Alzheimer’s disease, neuropathic pain, digestive sickness, cardiovascular illness, and cancer [21][22].
Also, a wide range of pharmacological activities of α- and β-pinene have been reported, such as anticoagulant, anti-inflammatory, anti-leishmania, antimalarial, antimicrobial, antioxidant, antitumor, analgesic, and antibiotic resistance modulation effects [23].
Camphene has several biological properties including anti-fungal, anti-inflammatory, analgesic, and antioxidant. Furthermore, camphene has been reported to exhibit a hypolipidemic action and it is also used as an expectorant and antinociceptive [24].
Camphor has several biological properties including anti-fungal, anti-microbial, anti-inflammatory, antioxidant, and analgesic [25]. Borneol exhibits a variety of biological activities, including neuroprotective, anti-inflammatory, antioxidant, antibacterial, analgesic, and drug penetration-enhancing effects [26].
Verbenone exhibits a wide spectrum of biological activities, including antihyperglycemic, antifungal, antibacterial, anti-inflammatory, and acaricidal effects [27].
It was observed that certain compounds were present in the essential oil of organically grown rosemary (α-myrcene, λ-terpinene, α-caryophyllene and Delta-cadinene), while these compounds were absent in the essential oil of conventionally grown rosemary, which contained compounds (tricyclene, β-thujene, β-myrcene, Delta-3-carene, cymene, p-cymene, thujone, sabinol, pinocamphone, iso-pinocamphone, verbenone, butyric acid-hexyl ester, safranal, terpinene acetate and cis-α-copaene-8-ol) that were not found in the organic plant.
For antioxidant activity.
When the free radical DPPH• interacts with an unpaired electron, maximum absorption occurs at 517 nm (purple color). An antioxidant that scavenges free radicals reacts with DPPH• to form DPPHH, which exhibits lower absorbance than DPPH due to a reduced amount of hydrogen. In comparison to the DPPH-H state, this radical form leads to decolorization (appearing yellow) as the number of collected electrons increases.
Ascorbic acid was used as a standard for the construction of a calibration curve. IC50 value was calculated from the calibration curve Y = 1.596x – 1.8517 with R2 = 0.9982, (where: x is the RSA, and Y is the concentration of ascorbic acid). IC50 = 32.489 ± 0.84 (µg/ml)
Also, gallic acid was used as a standard for the construction of a calibration curve. IC50 value was calculated from the calibration curve Y = 4.0612x + 1.1198 with R2 = 0.997, (where: x is the RSA, Y is the concentration of gallic acid). IC50 = 12.036 ± 0.51 (µg/ml)
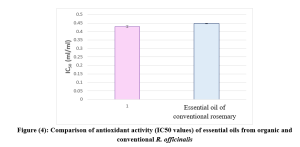
The antioxidant activities of R. officinalis essential oils were evaluated and expressed as the percentage of DPPH• radical inhibition. The concentrations and free radical scavenging (RSA%) for each essential oil are shown in (Figure 3).
The IC50 (Inhibitory Concentration of 50% of free radical DPPH•) for each EO was calculated based on the linear equation derived from the graphical representation of the relationship between the concentrations of the EO and their corresponding RSA values. The IC50 values calculated as the mean ± SD from triplicate experiments.
IC50 values of (0.429 ± 0.006 ml/ml) for organically grown rosemary, and (0.447 ± 0.002 ml/ml) for conventionally grown rosemary (Figure 4). Generally, the compounds most responsible for neutralizing DPPH• radicals were oxygenated monoterpenes, along with a monoterpene hydrocarbon.
The antioxidant activity of rosemary essential oil is attributed to several compounds, including 1,8-cineol, α-pinene, camphor, and camphene. These components are known for their ability to scavenge free radicals. Additionally, terpineol is also mentioned as contributing to the antioxidant properties of rosemary oil. According to our results, the oxygenated compounds were higher in the organic sample compared to the conventional sample, and it was observed that there is a difference in the percentage of certain compounds between samples grown organically and those grown conventionally. Additionally, some compounds appeared in one type but not in the other. The essential oil of organically cultivated rosemary showed a free radical scavenging activity comparable to the conventionally cultivated rosemary. The antioxidant capacity of plants enhances its significance in the food, cosmetic, and pharmaceutical industries, where its natural compounds could serve as effective alternatives to synthetic preservatives and antioxidants, as food chemical antioxidants have shown protective effects against reactive oxygen species and free radicals, but when present in excess, they may have harmful effects on health. Therefore, replacing these synthetic additives with non-toxic natural antioxidants is crucial [25].
In summary, the variations observed in phytochemical contents between organic and conventional Rosmarinus officinalis essential oil can be explained by several factors. Organic cultivation induces mild environmental stress and promotes microbial diversity in the soil, which stimulates the production of secondary metabolites as natural defense compounds. Meanwhile, differences in fertilization and pesticide use influence metabolic pathways, resulting in distinct chemical profiles. These insights highlight the complex interactions between cultivation practices and plant bioactive compounds [27].
Conclusions
This study is the first effect report of organic cultivation on the yield, chemical composition, and antioxidant activity of R. officinalis essential oil. According to our in vitro results, the antioxidant activity of the essential oil of R. officinalis were not affected by the type of agriculture. On the other hand, organic agriculture improved the yield of essential oil and enhanced the concentration of certain bioactive compounds of rosemary essential oil, which may have implications for its efficacy in pharmaceutical, cosmetic, and aromatherapy applications.
List of abbreviations
EOs: Essential oils, GC-MS: gas chromatography – mass spectrometry, RSA: Radical scavenging activity, DPPH•: 2,2-diphenyl -1- picrylhydrazyl, IC50: Inhibitory Concentration of 50% of free radical DPPH•, R.O.O: Essential oil of organic rosemary, R.C.O: Essential oil of conventional rosemary, RT: Retention time.
Ethics approval and consent to participate
Not applicable
Conflict of Interests
The authors declare no conflicts of interest.
Authors’ contributions
Abeer Mourda: Writing – original draft, Data curation, Methodology, Resources. Mays Khazem: Writing – review & editing, Supervision, Methodology, Resources. Jalal Fandi: Supervision, Methodology, Resources.
المراجع :Mena P, Cirlini M, Tassotti M, Herrlinger KA, Dall’Asta C, Del Rio D. Phytochemical profiling of flavonoids, phenolic acids, terpenoids, and volatile fraction of a rosemary (Rosmarinus officinalis L.) extract. Molecules. 2016. vol:21. iss:11. pp:1–15.
2. Nunziata A, De Benedetti L, Marchioni I, Cervelli C. High throughput measure of diversity in cytoplasmic and nuclear traits for unravelling geographic distribution of rosemary. Ecol Evol. 2019. vol:9. iss:7. pp:3728–3739.
3. Elyemni M, Louaste B, Nechad I, Elkamli T, Bouia A, Taleb M, et al. Extraction of Essential Oils of Rosmarinus officinalis L. by Two Different Methods: Hydrodistillation and Microwave Assisted Hydrodistillation. Sci World J. 2019. vol:2019.
4. Sirocchi V, Devlieghere F, Peelman N, Sagratini G, Maggi F, Vittori S, et al. Effect of Rosmarinus officinalis L. essential oil combined with different packaging conditions to extend the shelf life of refrigerated beef meat. Food Chem. 2017. vol:221. pp:1069–1076.
5. Dehghani Bidgoli R, Azarnezhad N, Akhbari M, Ghorbani M. Salinity stress and PGPR effects on essential oil changes in Rosmarinus officinalis L. Agric Food Secur. 2019. vol:8. iss:1. pp:1–7.
6. Satyal P, Jones TH, Lopez EM, McFeeters RL, Ali NAA, Mansi I, et al. Chemotypic characterization and biological activity of rosmarinus officinalis. Foods. 2017. vol:6. iss:3. pp:1–15.
7. Leporini M, Bonesi M, Loizzo MR, Passalacqua NG, Tundis R. The essential oil of salvia rosmarinus spenn. From Italy as a source of health-promoting compounds: Chemical profile and antioxidant and cholinesterase inhibitory activity. Plants. 2020. vol:9. iss:6. pp:1–13.
8. Mossa ATH, Afia SI, Mohafrash SMM, Abou-Awad BA. Rosemary essential oil nanoemulsion, formulation, characterization and acaricidal activity against the two-spotted spider mite Tetranychus urticae Koch (Acari: Tetranychidae). J Plant Prot Res. 2019. vol:59. iss:1. pp:102–112.
9. Nieto G, Ros G, Castillo J. Antioxidant and Antimicrobial Properties of Rosemary (Rosmarinus officinalis, L.): A Review. Medicines. 2018. vol:5. iss:3. pp:98.
10. Martínez AL, González-Trujano ME, Pellicer F, López-Muñoz FJ, Navarrete A. Antinociceptive effect and GC/MS analysis of Rosmarinus officinalis L. essential oil from its aerial parts. Planta Med. 2009. vol:75. iss:5. pp:508–511.
11. Shiwakoti S, Saleh O, Poudyal S, Barka A, Qian Y, Zheljazkov VD. Yield, Composition and Antioxidant Capacity of the Essential Oil of Sweet Basil and Holy Basil as Influenced by Distillation Methods. Chem Biodivers. 2017. vol:14. iss:4.
12. Moghaddam M, Miran SNK, Pirbalouti AG, Mehdizadeh L, Ghaderi Y. Variation in essential oil composition and antioxidant activity of cumin (Cuminum cyminum L.) fruits during stages of maturity. Ind Crops Prod. 2015. vol:70. pp:163–169.
13. Reganold JP, Wachter JM. Organic agriculture in the twenty-first century. Nat Plants. 2016. vol:2. iss:2. pp:1–8.
14. Forman J, Silverstein J, Bhatia JJS, Abrams SA, Corkins MR, De Ferranti SD, et al. Organic foods: Health and environmental advantages and disadvantages. Pediatrics. 2012. vol:130. iss:5.
15. Meemken EM, Qaim M. Organic Agriculture, Food Security, and the Environment. Annu Rev Resour Econ. 2018. vol:10. pp:39–63.
16. Lockeretz W. What Explains the Rise of Organic Farming? Organic Farming: an International History. 2007. 30–39 p.
17. Rosati A, Borek R, Canali S. Agroforestry and organic agriculture. Agrofor Syst. 2021. vol:95. iss:5. pp:805–821.
18. Bozin B, Mimica-Dukic N, Samojlik I, Jovin E. Antimicrobial and antioxidant properties of Rosemary and Sage (Rosmarinus officinalis L. and Salvia officinalis L., Lamiaceae) essential oils. J Agric Food Chem. 2007. vol:55. iss:19. pp:7879–7885.
19. Llauradó Maury G, Méndez Rodríguez D, Hendrix S, Escalona Arranz JC, Fung Boix Y, Pacheco AO, et al. Antioxidants in plants: A valorization potential emphasizing the need for the conservation of plant biodiversity in cuba. Antioxidants. 2020. vol:9. iss:11. pp:1–39.
20. Choi Y, Jeong HS, Lee J. Antioxidant activity of methanolic extracts from some grains consumed in Korea. Food Chem. 2007. vol:103. iss:1. pp:130–138.
21. Hoch CC, Petry J, Griesbaum L, Weiser T, Werner K, Ploch M, et al. 1,8-cineole (eucalyptol): A versatile phytochemical with therapeutic applications across multiple diseases. Biomed Pharmacother. 2023. vol:167.
22. Cai ZM, Peng JQ, Chen Y, Tao L, Zhang YY, Fu LY, et al. 1,8-Cineole: a review of source, biological activities, and application. J Asian Nat Prod Res. 2021. vol:23. iss:10. pp:938–954.
23. Park BB, An JY, Park SU. Letter to the editor : RECENT STUDIES ON PINENE AND ITS BIOLOGICAL AND PHARMACOLOGICAL ACTIVITIES Department of Environment and Forest Resources , Chungnam National University , * Corresponding author : Sang Un Park , Department of Crop Science , Chungnam. EXCLI J. 2021. iss:Table 1. pp:812–818.
24. Stamatiou R, Anagnostopoulou M, Ioannidou-Kabouri K, Rapti C, Lazou A. Camphene as a Protective Agent in Myocardial Ischemia/Reperfusion Injury. Antioxidants. 2024. vol:13. iss:4.
25. Duda-Madej A, Viscardi S, Grabarczyk M, Topola E, Kozłowska J, Mączka W, et al. Is Camphor the Future in Supporting Therapy for Skin Infections? Pharmaceuticals. 2024. vol:17. iss:6. pp:1–29.
26. Tang M, Zhong W, Guo L, Zeng H, Pang Y. Role of borneol as enhancer in drug formulation: A review. Chinese Herb Med. 2024. vol:17. iss:3. pp:473–483.
27. Mołdoch J, Agacka-Mołdoch M, Jóźwiak G, Wojtunik-Kulesza K. Biological Activity of Monoterpene-Based Scaffolds: A Natural Toolbox for Drug Discovery. Molecules. 2025. vol:30. iss:7.
28. El Maimouni MA, El Amrani S, Fadil M, Menyiy N, Bouslamti R, Annemer S, et al. Chemical Composition, Antioxidant Activity, and Multivariate Analysis of Four Moroccan Essential Oils: Mentha piperita, Mentha pulegium, Thymus serpyllum, and Thymus zygis. ScientificWorldJournal. 2024. vol:2024. pp:5552496.
29. Tarozzi A, Hrelia S, Angeloni C, Morroni F, Biagi P, Guardigli M, et al. Antioxidant effectiveness of organically and non-organically grown red oranges in cell culture systems. Eur J Nutr. 2006. vol:45. iss:3. pp:152–158.